3D Printing Hydrogel for Flexible Mechanical Sensors
DOI:
https://doi.org/10.64509/jim.11.43Keywords:
3D Printing, Hydrogel, Sensor, Health Monitoring, Human-Machine Interaction, Motion DetectionAbstract
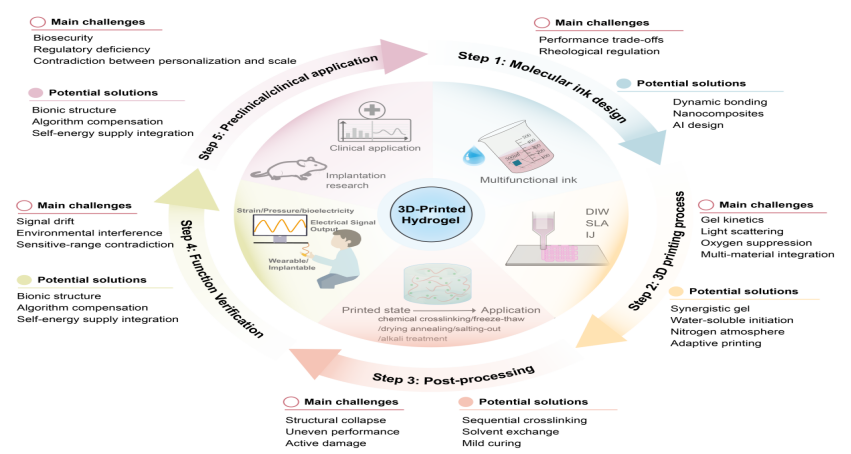
The growing demand for flexible sensors, driven by advances in flexible electronics, highlights hydrogels as an ideal material due to their intrinsic flexibility, biocompatibility, and multifunctional sensing capabilities. The fabrication of hydrogel devices with complex three-dimensional (3D) architectures and customized functions remains a challenge for traditional methods, while the limitation is now being overcome by 3D printing technology. Compared with 3D bioprinting, which has entered an advanced stage of development and 3D printing of plastics, metals, and resins, where technologies are relatively mature, 3D printing of hydrogel devices is still in the middle exploration stage. In this review, we mainly introduce recent progress on how to use 3D printing to build high-performance flexible electronic devices based on hydrogels, from the perspective of regulating key 3D printing process parameters. We emphasize the influence of material parameters, process parameters, and post-processing on device performance. We explain the working principles, performance indicators, and material characteristics of mainstream sensors such as resistive sensors, introduce various 3D printing methods for hydrogels, then discuss the ink design principles and the balance between printing quality and printing functions, and summarize the latest progress of these sensors in health monitoring, motion detection, and human-machine interaction. In short, this article looks forward to the future development direction of this attractive field.
Downloads
References
[1] Li, G., Li, C., Li, G., Yu, D., Song, Z., Wang, H., Liu, X., Liu, H., Liu, W.: Development of Conductive Hydrogels for Fabricating Flexible Strain Sensors. Small 18(5), 2101518 (2021). https://doi.org/10.1002/smll.202101518 DOI: https://doi.org/10.1002/smll.202101518
[2] Zhu, T., Ni, Y., Biesold, G.M., Cheng, Y., Ge, M., Li, H., Huang, J., Lin, Z., Lai, Y.: Recent advances in conductive hydrogels: classifications, properties, and applications. Chemical Society Reviews 52(2), 473–509 (2023). https://doi.org/10.1039/D2CS00173J DOI: https://doi.org/10.1039/D2CS00173J
[3] Zeng, X., Teng, L., Wang, X., Lu, T., Leng, W., Wu, X., Li, D., Zhong, Y., Sun, X., Zhu, S., et al.: Efficient multi-physical crosslinked nanocomposite hydrogel for a conformal strain and self-powered tactile sensor. Nano Energy 135, 110669 (2025). https://doi.org/10.1016/j.nanoen.2025.110669 DOI: https://doi.org/10.1016/j.nanoen.2025.110669
[4] Wu, X., Yang, Z., Dong, Y., Teng, L., Li, D., Han, H., Zhu, S., Sun, X., Zeng, Z., Zeng, X., et al.: A Self-Powered, Skin Adhesive, and Flexible Human–Machine Interface Based on Triboelectric Nanogenerator. Nanomaterials 14(16), 1365 (2024). https://doi.org/10.3390/nano14161365 DOI: https://doi.org/10.3390/nano14161365
[5] Shi, Z., Meng, L., Shi, X., Li, H., Zhang, J., Sun, Q., Liu, X., Chen, J., Liu, S.: Morphological Engineering of Sensing Materials for Flexible Pressure Sensors and Artificial Intelligence Applications. NanoMicro Letters 14(1), 141 (2022). https://doi.org/10.1007/s40820-022-00874-w DOI: https://doi.org/10.1007/s40820-022-00874-w
[6] Chen, S., Zhang, Y., Li, Y., Wang, P., Hu, D.: Recent development of flexible force sensors with multiple environmental adaptations. Nano Energy 124, 109443 (2024). https://doi.org/10.1016/j.nanoen.2024.109443 DOI: https://doi.org/10.1016/j.nanoen.2024.109443
[7] Peng, S., Yu, Y., Wu, S., Wang, C.-H.: Conductive Polymer Nanocomposites for Stretchable Electronics: Material Selection, Design, and Applications. ACS Applied Materials & Interfaces 13(37), 43831–43854 (2021). https://doi.org/10.1021/acsami.1c15014 DOI: https://doi.org/10.1021/acsami.1c15014
[8] Gong, S., Lu, Y., Yin, J., Levin, A., Cheng, W.: Materials-Driven Soft Wearable Bioelectronics for Connected Healthcare. Chemical Reviews 124(2), 455–553 (2024). https://doi.org/10.1021/acs.chemrev.3c00502 DOI: https://doi.org/10.1021/acs.chemrev.3c00502
[9] Luo, Y., Abidian, M.R., Ahn, J.-H., Akinwande, D., Andrews, A.M., Antonietti, M., Bao, Z., Berggren, M., Berkey, C.A., Bettinger, C.J., et al.: Technology Roadmap for Flexible Sensors. ACS Nano 17(6), 5211–5295 (2023). https://doi.org/10.1021/acsnano.2c12606 DOI: https://doi.org/10.1021/acsnano.2c12606
[10] Raeisi, A., Farjadian, F.: Commercial hydrogel product for drug delivery based on route of administration. Frontiers in Chemistry 12, 1336717 (2024). https://doi.org/10.3389/fchem.2024.1336717 DOI: https://doi.org/10.3389/fchem.2024.1336717
[11] Dey, K., Agnelli, S., Sartore, L.: Designing Viscoelastic Gelatin-PEG Macroporous Hybrid Hydrogel with Anisotropic Morphology and Mechanical Properties for Tissue Engineering Application. Micro 3(2), 434–457 (2023). https://doi.org/10.3390/micro3020029 DOI: https://doi.org/10.3390/micro3020029
[12] Liang, Y., He, J., Guo, B.: Functional Hydrogels as Wound Dressing to Enhance Wound Healing. ACS Nano 15(8), 12687–12722 (2021). https://doi.org/10.1021/acsnano.1c04206 DOI: https://doi.org/10.1021/acsnano.1c04206
[13] Amirthalingam, S., Rajendran, A.K., Moon, Y.G., Hwang, N.S.: Stimuli-responsive dynamic hydrogels: design, properties and tissue engineering applications. Materials Horizons 10(9), 3325–3350 (2023). https://doi.org/10.1039/D3MH00399J DOI: https://doi.org/10.1039/D3MH00399J
[14] Cheng, L., Suresh K, S., He, H., Rajput, R.S., Feng, Q., Ramesh, S., Wang, Y., Krishnan, S., Ostrovidov, S., Camci-Unal, G., et al.: 3D Printing of Micro- and Nanoscale Bone Substitutes: A Review on Technical and Translational Perspectives. International Journal of Nanomedicine 16, 4289–4319 (2021). https://doi.org/10.2147/ijn.s311001 DOI: https://doi.org/10.2147/IJN.S311001
[15] Murab, S., Gupta, A., Włodarczyk-Biegun, M.K., Kumar, A., van Rijn, P., Whitlock, P., Han, S.S., Agrawal, G.: Alginate based hydrogel inks for 3D bioprinting of engineered orthopedic tissues. Carbohydrate Polymers 296, 119964 (2022). https://doi.org/10.1016/j.carbpol.2022.119964 DOI: https://doi.org/10.1016/j.carbpol.2022.119964
[16] Zhu, Z., Ng, D.W.H., Park, H.S., McAlpine, M.C.: 3D-printed multifunctional materials enabled by artificial-intelligence-assisted fabrication technologies. Nature Reviews Materials 6, 27–47 (2021). https://doi.org/10.1038/s41578-020-00235-2 DOI: https://doi.org/10.1038/s41578-020-00235-2
[17] Seoane-Viano, I., Januskaite, P., Alvarez-Lorenzo, C., Basit, A.W., Goyanes, A.: Semi-solid extrusion 3D printing in drug delivery and biomedicine: Personalised solutions for healthcare challenges. Journal of Controlled Release 332, 367–389 (2021). https://doi.org/10.1016/j.jconrel.2021.02.027 DOI: https://doi.org/10.1016/j.jconrel.2021.02.027
[18] Sun, J., Liu, Q., Yang, J.: Additive manufacturing of highly entangled polymer networks. Science 385(6708), 566–572 (2024). https://doi.org/10.1126/science.adn6925
[19] Li, J., Cao, J., Bian, R., Wan, R., Zhu, X., Lu, B., Gu, G.: Multimaterial cryogenic printing of three-dimensional soft hydrogel machines. Nature Communications 16(1), 185 (2025). https://doi.org/10.1038/s41467-024-55323-6 DOI: https://doi.org/10.1038/s41467-024-55323-6
[20] Ji, Y., Su, E., Yee, D.W.: Volumetric Additive Manufacturing of Composites via Hydrogel Infusion. ACS Materials Letters 7(8), 2850–2857 (2025). https://doi.org/10.1021/acsmaterialslett.5c00407 DOI: https://doi.org/10.1021/acsmaterialslett.5c00407
[21] Daly, A.C., Davidson, M.D., Burdick, J.A.: 3D bioprinting of high cell-density heterogeneous tissue models through spheroid fusion within self-healing hydrogels. Nature Communications 12(1), 753 (2021). https://doi.org/10.1038/s41467-021-21029-2 DOI: https://doi.org/10.1038/s41467-021-21029-2
[22] Lin, X., Teng, Y., Xue, H., Bing, Y., Li, F., Wang, J., Li, J., Zhao, H., Zhang, T.: Janus Conductive Mechanism: An Innovative Strategy Enabling Ultra-Wide Linearity Range Pressure Sensing for Multi-Scenario Applications. Advanced Functional Materials 34(32), 2316314 (2024). https://doi.org/10.1002/adfm.202316314 DOI: https://doi.org/10.1002/adfm.202316314
[23] Li, G., Chu, Z., Gong, X., Xiao, M., Dong, Q., Zhao, Z., Hu, T., Zhang, Y., Wang, J., Tan, Y., et al.: A Wide-Range Linear and Stable Piezoresistive Sensor Based on Methylcellulose-Reinforced, Lamellar, and Wrinkled Graphene Aerogels. Advanced Materials Technologies 7(5), 2101021 (2021). https://doi.org/10.1002/admt.202101021 DOI: https://doi.org/10.1002/admt.202101021
[24] Zhu, G., Dai, H., Yao, Y., Tang, W., Shi, J., Yang, J., Zhu, L.: 3D Printed Skin-Inspired Flexible Pressure Sensor with Gradient Porous Structure for Tunable High Sensitivity and Wide Linearity Range. Advanced Materials Technologies 7(7), 2101239 (2021). https://doi.org/10.1002/admt.202101239 DOI: https://doi.org/10.1002/admt.202101239
[25] Shu, Q., Pang, Y., Li, Q., Gu, Y., Liu, Z., Liu, B., Li, J., Li, Y.: Flexible resistive tactile pressure sensors. Journal of Materials Chemistry A 12(16), 9296–9321 (2024). https://doi.org/10.1039/D3TA06976A DOI: https://doi.org/10.1039/D3TA06976A
[26] Wackers, G., Putzeys, T., Peeters, M., Van de Cauter, L., Cornelis, P., W¨ubbenhorst, M., Tack, J., Troost, F., Verhaert, N., Doll, T., et al.: Towards a catheter-based impedimetric sensor for the assessment of intestinal histamine levels in IBS patients. Biosensors and Bioelectronics 158, 112152 (2020). https://doi.org/10.1016/j.bios.2020.112152 DOI: https://doi.org/10.1016/j.bios.2020.112152
[27] Zhao, S., Liu, D., Yan, F.: Wearable Resistive-Type Stretchable Strain Sensors: Materials and Applications. Advanced Materials 37(5), 2413929 (2024). https://doi.org/10.1002/adma.202413929 DOI: https://doi.org/10.1002/adma.202413929
[28] Hu, R., Liu, P., Zhu, W., Guo, X., Liu, Z., Jin, Z., Yang, R., Han, J., Tian, H., Ma, Y., et al.: Design, Fabrication, and Wearable Medical Application of a High-Resolution Flexible Capacitive Temperature Sensor Based on the Thermotropic Phase Transition Composites of PEO/PVDF-HFP/H3PO4. Advanced Functional Materials 35(11), 2417205 (2024). https://doi.org/10.1002/adfm.202417205 DOI: https://doi.org/10.1002/adfm.202417205
[29] Qin, J., Yin, L.J., Hao, Y.N., Zhong, S.L., Zhang, D.L., Bi, K., Zhang, Y.X., Zhao, Y., Dang, Z.M.: Flexible and Stretchable Capacitive Sensors with Different Microstructures. Advanced Materials 33(34), 2008267 (2021). https://doi.org/10.1002/adma.202008267 DOI: https://doi.org/10.1002/adma.202008267
[30] Tay, R.Y., Li, H., Lin, J., Wang, H., Lim, J.S.K., Chen, S., Leong, W.L., Tsang, S.H., Teo, E.H.T.: Lightweight, Superelastic Boron Nitride/Polydimethylsiloxane Foam as Air Dielectric Substitute for Multifunctional Capacitive Sensor Applications. Advanced Functional Materials 30(10), 1909604 (2020). https://doi.org/10.1002/adfm.201909604 DOI: https://doi.org/10.1002/adfm.201909604
[31] Ma, Z., Zhang, Y., Zhang, K., Deng, H., Fu, Q.: Recent progress in flexible capacitive sensors: Structures and properties. Nano Materials Science 5(3), 265–277 (2023). https://doi.org/10.1016/j.nanoms.2021.11.002 DOI: https://doi.org/10.1016/j.nanoms.2021.11.002
[32] Basarir, F., Haj, Y.A., Zou, F., De, S., Nguyen, A., Frey, A., Haider, I., Sariola, V., Vapaavuori, J.: Edible and Biodegradable Wearable Capacitive Pressure Sensors: A Paradigm Shift toward Sustainable Electronics with Bio-Based Materials. Advanced Functional Materials 34(39), 2403268 (2024). https://doi.org/10.1002/adfm.202403268 DOI: https://doi.org/10.1002/adfm.202403268
[33] Qin, R., Hu, M., Li, X., Liang, T., Tan, H., Liu, J., Shan, G.: A new strategy for the fabrication of a flexible and highly sensitive capacitive pressure sensor. Microsystems & Nanoengineering 7(1), 100 (2021). https://doi.org/10.1038/s41378-021-00327-1 DOI: https://doi.org/10.1038/s41378-021-00327-1
[34] Basarir, F., Madani, Z., Vapaavuori, J.: Recent Advances in Silver Nanowire Based Flexible Capacitive Pressure Sensors: From Structure, Fabrication to Emerging Applications. Advanced Materials Interfaces 9(31), 2200866 (2022). https://doi.org/10.1002/admi.202200866 DOI: https://doi.org/10.1002/admi.202200866
[35] Mariello, M., Fachechi, L., Guido, F., De Vittorio, M.: Conformal, Ultra-thin Skin-Contact-Actuated Hybrid Piezo/Triboelectric Wearable Sensor Based on AlN and Parylene-Encapsulated Elastomeric Blend. Advanced Functional Materials 31(27), 2101047 (2021). https://doi.org/10.1002/adfm.202101047 DOI: https://doi.org/10.1002/adfm.202101047
[36] Li, Y., Luo, Y., Deng, H., Shi, S., Tian, S., Wu, H., Tang, J., Zhang, C., Zhang, X., Zha, J.W., et al.: Advanced Dielectric Materials for Triboelectric Nanogenerators: Principles, Methods, and Applications. Advanced Materials 36(52), 2314380 (2024). https://doi.org/10.1002/adma.202314380 DOI: https://doi.org/10.1002/adma.202314380
[37] Dassanayaka, D.G., Alves, T.M., Wanasekara, N.D., Dharmasena, I.G., Ventura, J.: Recent Progresses in Wearable Triboelectric Nanogenerators. Advanced Functional Materials 32(44), 2205438 (2022). https://doi.org/10.1002/adfm.202205438 DOI: https://doi.org/10.1002/adfm.202205438
[38] Deng, W., Yang, T., Jin, L., Yan, C., Huang, H., Chu, X., Wang, Z., Xiong, D., Tian, G., Gao, Y., et al.: Cowpea-structured PVDF/ZnO nanofibers based flexible self-powered piezoelectric bending motion sensor towards remote control of gestures. Nano Energy 55, 516–525 (2019). https://doi.org/10.1016/j.nanoen.2018.10.049 DOI: https://doi.org/10.1016/j.nanoen.2018.10.049
[39] Yang, L., Liu, Q., Li, M., Liu, Y., Li, X., Liu, Q., Zhu, T., Lu, Y., Liu, X., Wang, D.: A high-performance, flexible, and dual-modal humidity-piezoelectric sensor without mutual interference. Sensors and Actuators B: Chemical 423, 136778 (2025). https://doi.org/10.1016/j.snb.2024.136778 DOI: https://doi.org/10.1016/j.snb.2024.136778
[40] Chen, Y., Qin, C., Sun, Q., Wang, M.: Arrayed multilayer piezoelectric sensor based on electrospun P(VDF-TrFE)/ZnO with enhanced piezoelectricity. Sensors and Actuators A: Physical 379, 115970 (2024). https://doi.org/10.1016/j.sna.2024.115970 DOI: https://doi.org/10.1016/j.sna.2024.115970
[41] Cao, C., Zhou, P., Qin, W., Wang, J., Liu, M., Wang, P., Zhang, T., Qi, Y.: Enhanced sensing performance of piezoelectric pressure sensors via integrated nanofillers incorporated with P(VDF-TrFE) composites. Applied Surface Science 685, 162028 (2025). https://doi.org/10.1016/j.apsusc.2024.162028 DOI: https://doi.org/10.1016/j.apsusc.2024.162028
[42] Zhen, L., Cui, M., Bai, X., Jiang, J., Ma, X., Wang, M., Liu, J., Yang, B.: Thin, flexible hybrid-structured piezoelectric sensor array with enhanced resolution and sensitivity. Nano Energy 131, 110188 (2024). https://doi.org/10.1016/j.nanoen.2024.110188 DOI: https://doi.org/10.1016/j.nanoen.2024.110188
[43] Yin, B., Gosecka, M., Bodaghi, M., Crespy, D., Youssef, G., Dodda, J.M., Wong, S.H.D., Imran, A.B., Gosecki, M., Jobdeedamrong, A., et al.: Engineering multifunctional dynamic hydrogel for biomedical and tissue regenerative applications. Chemical Engineering Journal 487, 150403 (2024). https://doi.org/10.1016/j.cej.2024.150403 DOI: https://doi.org/10.1016/j.cej.2024.150403
[44] Zhang, Y., Tan, Y., Lao, J., Gao, H., Yu, J.: Hydrogels for Flexible Electronics. ACS Nano 17(11), 9681–9693 (2023). https://doi.org/10.1021/acsnano.3c02897 DOI: https://doi.org/10.1021/acsnano.3c02897
[45] Li, H., Cao, J., Wan, R., Feig, V.R., Tringides, C.M., Xu, J., Yuk, H., Lu, B.: PEDOTs-Based Conductive Hydrogels: Design, Fabrications, and Applications. Advanced Materials 37(7), 2415151 (2024). https://doi.org/10.1002/adma.202415151 DOI: https://doi.org/10.1002/adma.202415151
[46] Fatih, P., Karen, L.: 3D Printing of Polymer Hydrogels-From Basic Techniques to Programmable Actuation. Advanced Functional Materials 32(39), 2205345 (2022). https://doi.org/10.1002/adfm.202205345 DOI: https://doi.org/10.1002/adfm.202205345
[47] He, X., Zhang, B., Liu, Q., Chen, H., Cheng, J., Jian, B., Yin, H., Li, H., Duan, K., Zhang, J., et al.: Highly conductive and stretchable nanostructured ionogels for 3D printing capacitive sensors with superior performance. Nature Communications 15(1), 6431 (2024). https://doi.org/10.1038/s41467-024-50797-w DOI: https://doi.org/10.1038/s41467-024-50797-w
[48] Dhand, A.P., Davidson, M.D., Burdick, J.A.: Lithography-based 3D printing of hydrogels. Nature Reviews Bioengineering 3(2), 108–125 (2024). https://doi.org/10.1038/s44222-024-00251-9 DOI: https://doi.org/10.1038/s44222-024-00251-9
[49] Zhu, Z., Park, H.S., McAlpine, M.C.: 3D printed deformable sensors. Science Advances 6(25), eaba5575 (2020). https://doi.org/10.1126/sciadv.aba5575 DOI: https://doi.org/10.1126/sciadv.aba5575
[50] Sun, Y., Kumar, V., Adesida, I., Rogers, J.A.: Buckled and Wavy Ribbons of GaAs for High-Performance Electronics on Elastomeric Substrates. Advanced Materials 18(21), 2857–2862 (2006). https://doi.org/10.1002/adma.200600646 DOI: https://doi.org/10.1002/adma.200600646
[51] Mannsfeld, S.C.B., Tee, B.C.K., Stoltenberg, R.M., Chen, C.V.H.H., Barman, S., Muir, B.V.O., Sokolov, A.N., Reese, C., Bao, Z.: Highly sensitive flexible pressure sensors with microstructured rubber dielectric layers. Nature Materials 9(10), 859–864 (2010). https://doi.org/10.1038/nmat2834 DOI: https://doi.org/10.1038/nmat2834
[52] Lipomi, D.J., Vosgueritchian, M., Tee, B.C.K., Hellstrom, S.L., Lee, J.A., Fox, C.H., Bao, Z.: Skin-like pressure and strain sensors based on transparent elastic films of carbon nanotubes. Nature Nanotechnology 6(12), 788–792 (2011). https://doi.org/10.1038/nnano.2011.184 DOI: https://doi.org/10.1038/nnano.2011.184
[53] Fan, F.-R., Lin, L., Zhu, G., Wu, W., Zhang, R., Wang, Z.L.: Transparent Triboelectric Nanogenerators and Self-Powered Pressure Sensors Based on Micropatterned Plastic Films. Nano Letters 12(6), 3109–3114 (2012). https://doi.org/10.1021/nl300988z DOI: https://doi.org/10.1021/nl300988z
[54] Chun, J., Lee, K.Y., Kang, C.Y., Kim, M.W., Kim, S.W., Baik, J.M.: Embossed Hollow Hemisphere-Based Piezoelectric Nanogenerator and Highly Responsive Pressure Sensor. Advanced Functional Materials 24(14), 2038–2043 (2013). https://doi.org/10.1002/adfm.201302962 DOI: https://doi.org/10.1002/adfm.201302962
[55] Shao, Q., Niu, Z., Hirtz, M., Jiang, L., Liu, Y., Wang, Z., Chen, X.: High-Performance and Tailorable Pressure Sensor Based on Ultrathin Conductive Polymer Film. Small 10(8), 1466–1472 (2014). https://doi.org/10.1002/smll.201303601 DOI: https://doi.org/10.1002/smll.201303601
[56] Lee, J., Kwon, H., Seo, J., Shin, S., Koo, J.H., Pang, C., Son, S., Kim, J.H., Jang, Y.H., Kim, D.E., et al.: Conductive Fiber-Based Ultrasensitive Textile Pressure Sensor for Wearable Electronics. Advanced Materials 27(15), 2433–2439 (2015). https://doi.org/10.1002/adma.201500009 DOI: https://doi.org/10.1002/adma.201500009
[57] Wang, Z., Wang, S., Zeng, J., Ren, X., Chee, A.J.Y., Yiu, B.Y.S., Chung, W.C., Yang, Y., Yu, A.C.H., Roberts, R.C., et al.: High Sensitivity, Wearable, Piezoresistive Pressure Sensors Based on Irregular Microhump Structures and Its Applications in Body Motion Sensing. Small 12(28), 3827–3836 (2016). https://doi.org/10.1002/smll.201601419 DOI: https://doi.org/10.1002/smll.201601419
[58] Luo, N., Huang, Y., Liu, J., Chen, S.C., Wong, C.P., Zhao, N.: Hollow-Structured Graphene–Silicone-Composite-Based Piezoresistive Sensors: Decoupled Property Tuning and Bending Reliability. Advanced Materials 29(40), 1702675 (2017). https://doi.org/10.1002/adma.201702675 DOI: https://doi.org/10.1002/adma.201702675
[59] Chun, S., Son, W., Choi, C.: Flexible pressure sensors using highly-oriented and free-standing carbon nanotube sheets. Carbon 139, 586–592 (2018). https://doi.org/10.1016/j.carbon.2018.07.005 DOI: https://doi.org/10.1016/j.carbon.2018.07.005
[60] Chang, Q., Darabi, M.A., Liu, Y., He, Y., Zhong, W., Mequanin, K., Li, B., Lu, F., Xing, M.M.Q.: Hydrogels from natural egg white with extraordinary stretchability, direct-writing 3D printability and self-healing for fabrication of electronic sensors and actuators. Journal of Materials Chemistry A 7(42), 24626–24640 (2019). https://doi.org/10.1039/C9TA06233E DOI: https://doi.org/10.1039/C9TA06233E
[61] Wu, Y., Zeng, Y., Chen, Y., Li, C., Qiu, R., Liu, W.: Photocurable 3D Printing of High Toughness and Self-Healing Hydrogels for Customized Wearable Flexible Sensors. Advanced Functional Materials 31(52), 2107202 (2021). https://doi.org/10.1002/adfm.202107202 DOI: https://doi.org/10.1002/adfm.202107202
[62] Haodong, L., Chunyu, D., Liling, L., Hongjian, Z., Haiqing, Z., Weichang, Z., Tianning, R., Zhicheng, S., Yuquan, L., Zhentao, N., et al.: Approaching intrinsic dynamics of MXenes hybrid hydrogel for 3D printed multimodal intelligent devices with ultrahigh superelasticity and temperature sensitivity. Nature Communications 13(1), 3420 (2022). https://doi.org/10.1038/s41467-022-31051-7 DOI: https://doi.org/10.1038/s41467-022-31051-7
[63] Jingxia, Z., Guoqi, C., Hongjuan, Y., Canjie, Z., Shengnan, L., Wenquan, W., Jiangtao, R., Yang, C., Xun, X., Xinwei, W., et al.: 3D printed microstructured ultrasensitive pressure sensors based on microgel-reinforced double network hydrogels for biomechanical applications. Materials Horizons 10(10), 4232–4242 (2023). https://doi.org/10.1039/D3MH00718A DOI: https://doi.org/10.1039/D3MH00718A
[64] Tang, H., Yang, Y., Liu, Z., Li, W., Zhang, Y., Huang, Y., Kang, T., Yu, Y., Li, N., Tian, Y., et al.: Injectable ultrasonic sensor for wireless monitoring of intracranial signals. Nature 630(8015), 84–90 (2024). https://doi.org/10.1038/s41586-024-07334-y DOI: https://doi.org/10.1038/s41586-024-07334-y
[65] Zhou, X., Yu, X., You, T., Zhao, B., Dong, L., Huang, C., Zhou, X., Xing, M., Qian, W., Luo, G.: 3D Printing-Based Hydrogel Dressings for Wound Healing. Advanced Science 11(47), 2404580 (2024). https://doi.org/10.1002/advs.202404580 DOI: https://doi.org/10.1002/advs.202404580
[66] Vijayavenkataraman, S., Yan, W.-C., Lu, W.F., Wang, C.-H., Fuh, J.Y.H.: 3D bioprinting of tissues and organs for regenerative medicine. Advanced Drug Delivery Reviews 132, 296–332 (2018). https://doi.org/10.1016/j.addr.2018.07.004 DOI: https://doi.org/10.1016/j.addr.2018.07.004
[67] Ho, M., Ramirez, A.B., Akbarnia, N., Croiset, E., Prince, E., Fuller, G.G., Kamkar, M.: Direct Ink Writing of Conductive Hydrogels. Advanced Functional Materials 35(22), 2404580 (2025). https://doi.org/10.1002/adfm.202415507 DOI: https://doi.org/10.1002/adfm.202415507
[68] Baniasadi, H., Abidnejad, R., Fazeli, M., Lipponen, J., Niskanen, J., Kontturi, E., Seppala, J., Rojas, O.J.: Innovations in hydrogel-based manufacturing: A comprehensive review of direct ink writing technique for biomedical applications. Advances in Colloid and Interface Science 324, 103095 (2024). https://doi.org/10.1016/j.cis.2024.103095 DOI: https://doi.org/10.1016/j.cis.2024.103095
[69] Shahzad, A., Lazoglu, I.: Direct ink writing (DIW) of structural and functional ceramics: Recent achievements and future challenges. Composites Part B: Engineering 225, 109249 (2021). https://doi.org/10.1016/j.compositesb.2021.109249 DOI: https://doi.org/10.1016/j.compositesb.2021.109249
[70] Wu, Y., Zhang, Y., Yan, M., Hu, G., Li, Z., He, W., Wang, X., Abulimit, A., Li, R.: Research progress on the application of inkjet printing technology combined with hydrogels. Applied Materials Today 36, 102036 (2024). https://doi.org/10.1016/j.apmt.2023.102036 DOI: https://doi.org/10.1016/j.apmt.2023.102036
[71] Shah, M.A., Lee, D.-G., Lee, B.-Y., Hur, S.: Classifications and Applications of Inkjet Printing Technology: A Review. IEEE Access 9, 140079–140102 (2021). https://doi.org/10.1109/ACCESS.2021.3119219 DOI: https://doi.org/10.1109/ACCESS.2021.3119219
[72] Cheng, C., Williamson, E.J., Chiu, G.T.C., Han, B.: Engineering biomaterials by inkjet printing of hydrogels with functional particulates. Med-X 2(1), 9 (2024). https://doi.org/10.1007/s44258-024-00024-4 DOI: https://doi.org/10.1007/s44258-024-00024-4
[73] Teo, M.Y., Kee, S., RaviChandran, N., Stuart, L., Aw, K.C., Stringer, J.: Enabling Free-Standing 3D Hydrogel Microstructures with Microreactive Inkjet Printing. ACS Applied Materials & Interfaces 12(1), 1832–1839 (2019). https://doi.org/10.1021/acsami.9b17192 DOI: https://doi.org/10.1021/acsami.9b17192
[74] Quan, H., Zhang, T., Xu, H., Luo, S., Nie, J., Zhu, X.: Photo-curing 3D printing technique and its challenges. Bioactive Materials 5(1), 110–115 (2020). https://doi.org/10.1016/j.bioactmat.2019.12.003 DOI: https://doi.org/10.1016/j.bioactmat.2019.12.003
[75] Dhand, A.P., Davidson, M.D., Zlotnick, H.M., Kolibaba, T.J., Killgore, J.P., Burdick, J.A.: Additive manufacturing of highly entangled polymer networks. Science 385(6708), 566–572 (2024). https://doi.org/10.1126/science.adn6925 DOI: https://doi.org/10.1126/science.adn6925
[76] Ouyang, L.: Pushing the rheological and mechanical boundaries of extrusion-based 3D bioprinting. Trends in Biotechnology 40(7), 891–902 (2022). https://doi.org/10.1016/j.tibtech.2022.01.001 DOI: https://doi.org/10.1016/j.tibtech.2022.01.001
[77] Bedell, M.L., Navara, A.M., Du, Y., Zhang, S., Mikos, A.G.: Polymeric Systems for Bioprinting. Chemical Reviews 120(19), 10744–10792 (2020). https://doi.org/10.1021/acs.chemrev.9b00834 DOI: https://doi.org/10.1021/acs.chemrev.9b00834
[78] Piras, C.C., Smith, D.K.: Multicomponent polysaccharide alginate-based bioinks. Journal of Materials Chemistry B 8(36), 8171–8188 (2020). https://doi.org/10.1039/D0TB01005G DOI: https://doi.org/10.1039/D0TB01005G
[79] Hasany, M., Talebian, S., Sadat, S., Ranjbar, N., Mehrali, M., Wallace, G.G., Mehrali, M.: Synthesis, properties, and biomedical applications of alginate methacrylate (ALMA)-based hydrogels: Current advances and challenges. Applied Materials Today 24, 101150 (2021). https://doi.org/10.1016/j.apmt.2021.101150 DOI: https://doi.org/10.1016/j.apmt.2021.101150
[80] Lee, S., Choi, G., Yang, Y.J., Joo, K.I., Cha, H.J.: Visible light-crosslinkable tyramine-conjugated alginate-based microgel bioink for multiple cell-laden 3D artificial organ. Carbohydrate Polymers 313, 120895 (2023). https://doi.org/10.1016/j.carbpol.2023.120895 DOI: https://doi.org/10.1016/j.carbpol.2023.120895
[81] Agostinacchio, F., Fitzpatrick, V., Dir`e, S., Kaplan, D.L., Motta, A.: Silk fibroin-based inks for in situ 3D printing using a double crosslinking process. Bioactive Materials 35, 122–134 (2024). https://doi.org/10.1016/j.bioactmat.2024.01.015 DOI: https://doi.org/10.1016/j.bioactmat.2024.01.015
[82] Yan, X., Huang, H., Bakry, A.M., Wu, W., Liu, X., Liu, F.: Advances in enhancing the mechanical properties of biopolymer hydrogels via multi-strategic approaches. International Journal of Biological Macromolecules 272, 132583 (2024). https://doi.org/10.1016/j.ijbiomac.2024.132583 DOI: https://doi.org/10.1016/j.ijbiomac.2024.132583
[83] Liu, X., Hao, S., Wang, Y., Zheng, L., Hu, Z., Wen, J., Xu, F., Yang, J.: Mechanically adaptable microdomed hydrogels for flexible pressure sensors with broad detection range and stable signal output. Chemical Engineering Journal 505, 159113 (2025). https://doi.org/10.1016/j.cej.2024.159113 DOI: https://doi.org/10.1016/j.cej.2024.159113
[84] Yang, S., Pan, J., Fu, H., Zheng, J., Chen, F., Zhang, M., Gong, Z., Liang, K., Wang, C., Lai, J., et al.: Preparation of carbon-based conductive hydrogels and their potential for promoting nerve regeneration. Advanced Composites and Hybrid Materials 8(2), 185 (2025). https://doi.org/10.1007/s42114-025-01261-w DOI: https://doi.org/10.1007/s42114-025-01261-w
[85] Deng, W., Zhang, Y., Wu, M., Liu, C., Rahmaninia, M., Tang, Y., Li, B.: A tough, stretchable, adhesive and electroconductive polyacrylamide hydrogel sensor incorporated with sulfonated nanocellulose and carbon nanotubes. International Journal of Biological Macromolecules 279, 135165 (2024). https://doi.org/10.1016/j.ijbiomac.2024.135165 DOI: https://doi.org/10.1016/j.ijbiomac.2024.135165
[86] Liu, P., Yao, D., Lu, C., Gao, X., Dong, P.: Highly sensitive strain sensors based on PVA hydrogels with a conductive surface layer of graphene. Journal of Materials Science: Materials in Electronics 35(2), 97 (2024). https://doi.org/10.1007/s10854-024-11927-8 DOI: https://doi.org/10.1007/s10854-024-11927-8
[87] Zhou, T., Yuk, H., Hu, F., Wu, J., Tian, F., Roh, H., Shen, Z., Gu, G., Xu, J., Lu, B., et al.: 3D printable high-performance conducting polymer hydrogel for all-hydrogel bioelectronic interfaces. Nature Materials 22(7), 895–902 (2023). https://doi.org/10.1038/s41563-023-01569-2 DOI: https://doi.org/10.1038/s41563-023-01569-2
[88] GhavamiNejad, A., Ashammakhi, N., Wu, X.Y., Khademhosseini, A.: Crosslinking Strategies for 3D Bioprinting of Polymeric Hydrogels. Small 16(35), 2002931 (2020). https://doi.org/10.1002/smll.202002931 DOI: https://doi.org/10.1002/smll.202002931
[89] Zeng, L., Wang, J., Duan, L., Gao, G.: Highly transparent ionogel for wearable force sensor and 3D printing. European Polymer Journal 223, 113641 (2025). https://doi.org/10.1016/j.eurpolymj.2024.113641 DOI: https://doi.org/10.1016/j.eurpolymj.2024.113641
[90] Zhu, L., Rong, Y., Wang, Y., Bao, Q., An, J., Huang, D., Huang, X.: DLP printing of tough organogels for customized wearable sensors. European Polymer Journal 187, 111886 (2023). https://doi.org/10.1016/j.eurpolymj.2023.111886 DOI: https://doi.org/10.1016/j.eurpolymj.2023.111886
[91] Liu, S., Zhang, H., Ahlfeld, T., Kilian, D., Liu, Y., Gelinsky, M., Hu, Q.: Evaluation of different crosslinking methods in altering the properties of extrusion-printed chitosan-based multi-material hydrogel composites. Bio-Design and Manufacturing 6(2), 150–173 (2023). https://doi.org/10.1007/s42242-022-00194-3 DOI: https://doi.org/10.1007/s42242-022-00194-3
[92] Ouyang, L., Armstrong, J.P.K., Lin, Y., Wojciechowski, J.P., Lee-Reeves, C., Hachim, D., Zhou, K., Burdick, J.A., Stevens, M.M.: Expanding and optimizing 3D bioprinting capabilities using complementary network bioinks. Science Advances 6(38), eabc5529 (2020). https://doi.org/10.1126/sciadv.abc5529 DOI: https://doi.org/10.1126/sciadv.abc5529
[93] Zhang, X., Li, D., Liu, G.: 3D Printed Ultrasoft and Adhesive PEDOT:PSS-Based Hydrogel for Bioelectronics. ACS Applied Polymer Materials 7(3), 1531–1539 (2025). https://pubs.acs.org/doi/10.1021/acsapm.4c03275 DOI: https://doi.org/10.1021/acsapm.4c03275
[94] Choi, S., Lee, K.Y., Kim, S.L., MacQueen, L.A., Chang, H., Zimmerman, J.F., Jin, Q., Peters, M.M., Ardona, H.A.M., Liu, X., et al.: Fibre-infused gel scaffolds guide cardiomyocyte alignment in 3D-printed ventricles. Nature Materials 22(8), 1039–1046 (2023). https://doi.org/10.1038/s41563-023-01611-3 DOI: https://doi.org/10.1038/s41563-023-01611-3
[95] Guo, B., Lin, C., Ye, H., Xue, Y., Mo, J., Chen, J., Cui, Y., Fu, C., Bai, J., Ge, Q., et al.: 3D printed organohydrogel-based strain sensors with enhanced sensitivity and stability via structural design. International Journal of Extreme Manufacturing 7(5), 055507 (2025). https://doi.org/10.1088/2631-7990/add971 DOI: https://doi.org/10.1088/2631-7990/add971
[96] Wang, Y., Okuro, K.: Mechano-Chemiluminescent Hydrogel for Sustained Stress Visualization Under Mechanical Equilibrium. Macromolecular Rapid Communications 46(19), 2500256 (2025). https://doi.org/10.1002/marc.202500256 DOI: https://doi.org/10.1002/marc.70051
[97] Barrulas, R.V., Corvo, M.C.: Rheology in Product Development: An Insight into 3D Printing of Hydrogels and Aerogels. Gels 9(12), 986 (2023). https://doi.org/10.3390/gels9120986 DOI: https://doi.org/10.3390/gels9120986
[98] Cook-Chennault, K., Anaokar, S., Medina Vazquez, A.M., Chennault, M.: Influence of High Strain Dynamic Loading on HEMA–DMAEMA Hydrogel Storage Modulus and Time Dependence. Polymers 16(13), 1797 (2024). https://doi.org/10.3390/polym16131797 DOI: https://doi.org/10.3390/polym16131797
[99] Zhang, L., Bai, X., Liang, Y., Zhang, G., Zou, J., Lai, W., Fei, P.: Preparation of chitosan derivatives/oxidized carboxymethyl cellulose hydrogels by freeze-thaw method: Synthesis, characterization, and utilization in dye absorption. International Journal of Biological Macromolecules 282, 136924 (2024). https://doi.org/10.1016/j.ijbiomac.2024.136924 DOI: https://doi.org/10.1016/j.ijbiomac.2024.136924
[100] Liu, P., Zhang, Y., Guan, Y., Zhang, Y.: Peptide-Crosslinked, Highly Entangled Hydrogels with Excellent Mechanical Properties but Ultra-Low Solid Content. Advanced Materials 35(13), 2210021 (2023). https://doi.org/10.1002/adma.202210021 DOI: https://doi.org/10.1002/adma.202210021
[101] Podder, A.K., Mohamed, M.A., Seidman, R.A., Tseropoulos, G., Polanco, J.J., Lei, P., Sim, F.J., Andreadis, S.T.: Injectable shear-thinning hydrogels promote oligodendrocyte progenitor cell survival and remyelination in the central nervous system. Science Advances 10(28), eadk9918 (2024). https://doi.org/10.1126/sciadv.adk9918 DOI: https://doi.org/10.1126/sciadv.adk9918
[102] Brown, N.C., Ames, D.C., Mueller, J.: Multimaterial extrusion 3D printing printheads. Nature Reviews Materials 10, 807–825 (2025). https://doi.org/10.1038/s41578-025-00809-y DOI: https://doi.org/10.1038/s41578-025-00809-y
[103] Guvendiren, M., Lu, H.D., Burdick, J.A.: Shear-thinning hydrogels for biomedical applications. Soft Matter 8(2), 260–272 (2012). https://doi.org/10.1039/C1SM06513K DOI: https://doi.org/10.1039/C1SM06513K
[104] Yuan, Z., Huang, X., Zhang, X., Gao, S., Chen, H., Li, Z., El-Mesery, H.S., Shi, J., Zou, X.: Unveiling rheological behavior of hydrogels toward Magic 3D printing patterns. Food Hydrocolloids 168, 111505 (2025). https://doi.org/10.1016/j.foodhyd.2025.111505 DOI: https://doi.org/10.1016/j.foodhyd.2025.111505
[105] Kang, S.W., Mueller, J.: Multiscale 3D printing via active nozzle size and shape control. Science Advances 10(23), eadn7772 (2024). https://doi.org/10.1126/sciadv.adn7772 DOI: https://doi.org/10.1126/sciadv.adn7772
[106] Bono, F., Strassle Zuniga, S.H., Amstad, E.: 3D Printable k-Carrageenan-Based Granular Hydrogels. Advanced Functional Materials 35(3), 2413368 (2024). https://doi.org/10.1002/adfm.202413368 DOI: https://doi.org/10.1002/adfm.202413368
[107] Ge, Q., Chen, Z., Cheng, J., Zhang, B., Zhang, Y.-F., Li, H., He, X., Yuan, C., Liu, J., Magdassi, S., et al.: 3D printing of highly stretchable hydrogel with diverse UV curable polymers. Science Advances 7(2), eaba4261 (2021). https://doi.org/10.1126/sciadv.aba4261 DOI: https://doi.org/10.1126/sciadv.aba4261
[108] Highley, C.B., Rodell, C.B., Burdick, J.A.: Direct 3D Printing of Shear-Thinning Hydrogels into Self-Healing Hydrogels. Advanced Materials 27(34), 5075–5079 (2015). https://doi.org/10.1002/adma.201501234 DOI: https://doi.org/10.1002/adma.201501234
[109] Khan, K., Hussain, M.I., Tareen, A.K., Asghar, A., Hamza, M., Chen, Z.: Advances in vat photopolymerization 3D printing: Multifunctional materials, process innovations, and emerging applications. Materials Science and Engineering: R: Reports 167, 101120 (2026). https://doi.org/10.1016/j.mser.2025.101120 DOI: https://doi.org/10.1016/j.mser.2025.101120
[110] Wu, R., Zhu, T., Ni, Y., Wu, C., Wang, W., Zhao, K., Huang, J., Lai, Y.: UV-Cured Dense Double Network Hydrogel via Multiple Dynamic Crosslinking for Stable Amphibious Motion Sensing. Advanced Functional Materials 36(3), e15120 (2025). https://doi.org/10.1002/adfm.202515120 DOI: https://doi.org/10.1002/adfm.202515120
[111] Li, Q., Zhao, Y.L., Shen, H.X., Hou, Y.C., Lu, J.L., Zhu, L., Chen, S.: 3D Printing–In Situ Curing of Soft Organogels Using Frontal Polymerizable Inks. Advanced Materials 37(35), 2419039 (2025). https://doi.org/10.1002/adma.202419039 DOI: https://doi.org/10.1002/adma.202419039
[112] Ye, T., Chai, M., Wang, Z., Shao, T., Liu, J., Shi, X.: 3D-Printed Hydrogels with Engineered Nanocrystalline Domains as Functional Vascular Constructs. ACS Nano 18(37), 25765–25777 (2024). https://doi.org/10.1021/acsnano.4c08359 DOI: https://doi.org/10.1021/acsnano.4c08359
[113] Wang, Y., Xu, X., Liu, D., Wu, J., Yang, X., Zhu, B., Sun, C., Jiang, P., Wang, X.: Dimethyl sulfoxide mediated high-fidelity 3D printing of hydrogels. Additive Manufacturing 91, 104346 (2024). https://doi.org/10.1016/j.addma.2024.104346 DOI: https://doi.org/10.1016/j.addma.2024.104346
[114] Zhuo, S., Qian, Z., Sainan, M., Jingjun, W.: DLP 3D printed hydrogels with hierarchical structures post-programmed by lyophilization and ionic locking. Materials Horizons 10(1), 179–186 (2023). https://doi.org/10.1039/D2MH00962E DOI: https://doi.org/10.1039/D2MH00962E
[115] Cui, C., Zhuang, Z.Y., Gao, H.L., Pang, J., Pan, X.F., Yu, S.H.: 3D Printing of Ultrahigh Filler Content Composites Enabled by Granular Hydrogels. Advanced Materials 37(30), 2500782 (2025). https://doi.org/10.1002/adma.202500782 DOI: https://doi.org/10.1002/adma.202500782
[116] Wang, F., Xue, Y., Chen, X., Zhang, P., Shan, L., Duan, Q., Xing, J., Lan, Y., Lu, B., Liu, J.: 3D Printed Implantable Hydrogel Bioelectronics for Electrophysiological Monitoring and Electrical Modulation. Advanced Functional Materials 34(21), 2314471 (2023). https://doi.org/10.1002/adfm.202470116 DOI: https://doi.org/10.1002/adfm.202314471
[117] Wang, Z., Chen, L., Chen, Y., Liu, P., Duan, H., Cheng, P.: 3D Printed Ultrastretchable, Hyper-Antifreezing Conductive Hydrogel for Sensitive Motion and Electrophysiological Signal Monitoring. Research 2020, 1426078 (2020). https://doi.org/10.34133/2020/1426078 DOI: https://doi.org/10.34133/2020/1426078
[118] Zheng, S., Ruan, L., Meng, F., Wu, Z., Qi, Y., Gao, Y., Yuan, W.: Skin-Inspired, Multifunctional, and 3D-Printable Flexible Sensor Based on Triple-Responsive Hydrogel for Signal Conversion in Skin Interface Electronics Health Management. Small 21(6), 2408745 (2024). https://doi.org/10.1002/smll.202408745 DOI: https://doi.org/10.1002/smll.202408745
[119] Tang, J., Gou, K., Wang, C., Wei, M., Tan, Q., Weng, G.: Self-Powered and 3D Printable Soft Sensor for Human Health Monitoring, Object Recognition, and Contactless Hand Gesture Recognition. Advanced Functional Materials 34(52), 2411172 (2024). https://doi.org/10.1002/adfm.202411172 DOI: https://doi.org/10.1002/adfm.202411172
[120] Vera, D., Garcia-Diaz, M., Torras, N., Castillo, O., Illa, X., Villa, R., Alvarez, M., Martinez, E.: A 3D bioprinted hydrogel gut-on-chip with integrated electrodes for transepithelial electrical resistance (TEER) measurements. Biofabrication 16(3), 035008 (2024). https://doi.org/10.1088/1758-5090/ad3aa4 DOI: https://doi.org/10.1088/1758-5090/ad3aa4
[121] Hu, F., Yu, D., Dong, B., Gong, X., Li, Z., Zhao, R., Wang, Q., Li, G., Wang, H., Liu, W., et al.: Antibacterial conductive hydrogels with freeze-directed microstructures reinforced by polyaniline-encapsulated bacterial cellulose for flexible sensors. Chemical Engineering Journal 512, 162702 (2025). https://doi.org/10.1016/j.cej.2025.162702 DOI: https://doi.org/10.1016/j.cej.2025.162702
[122] Kim, H., Dutta, S.D., Jeon, M.J., Lee, J., Park, H., Seol, Y., Lim, K.T.: Bioinspired Shape Reconfigurable, Printable, and Conductive ”E-Skin” Patch with Robust Antibacterial Properties for Human Health Sensing. Advanced Functional Materials 35(40), 2504088 (2025). https://doi.org/10.1002/adfm.202504088 DOI: https://doi.org/10.1002/adfm.202504088
[123] Huang, D., Qi, J., Gao, S., Yin, L., Qi, H., Zheng, S.: DLP 3D-Printed Conic-Pyramid Hydrogel Sensor for Wearable Devices and Handwritten Fingerprint Recognition. ACS Applied Materials & Interfaces 17(33), 47273–47289 (2025). https://doi.org/10.1021/acsami.5c07889 DOI: https://doi.org/10.1021/acsami.5c07889
[124] Wu, K., Li, J., Li, Y., Wang, H., Zhang, Y., Guo, B., Yu, J., Wang, Y.: 3D Printed Silk Fibroin-Based Hydrogels with Tunable Adhesion and Stretchability for Wearable Sensing. Advanced Functional Materials 34(46), 2404451 (2024). https://doi.org/10.1002/adfm.202404451 DOI: https://doi.org/10.1002/adfm.202404451
[125] Zhang, S., Jia, W., Xu, T., Guo, A., Liu, Y., Liu, G., Yang, X., Liu, P., Yang, R., Sui, C.: High-sensitivity wearable sensor based on micro-needle structure design assisted by 3D printing. Chemical Engineering Journal 520, 165966 (2025). https://doi.org/10.1016/j.cej.2025.165966 DOI: https://doi.org/10.1016/j.cej.2025.165966
[126] Liu, Q., Dong, X., Qi, H., Zhang, H., Li, T., Zhao, Y., Li, G., Zhai, W.: 3D printable strong and tough composite organo-hydrogels inspired by natural hierarchical composite design principles. Nature Communications 15(1), 3237 (2024). https://doi.org/10.1038/s41467-024-47597-7 DOI: https://doi.org/10.1038/s41467-024-47597-7
[127] Li, J., Yu, Q., Shen, Z., Wang, D., Gu, G.: Soft 3D Lattice Iontronic Tactile Sensor. SmartBot 1(3), e70002 (2025). https://doi.org/10.1002/smb2.70002 DOI: https://doi.org/10.1002/smb2.70002
[128] Yang, J., Yang, K., An, X., Fan, Z., Li, Y., Yin, L., Long, Y., Pan, G., Liu, H., Ni, Y.: Highly Flexible, Stretchable, and Compressible Lignin-Based Hydrogel Sensors with Frost Resistance for Advanced Bionic Hand Control. Advanced Functional Materials 35(11), 2416916 (2025). https://doi.org/10.1002/adfm.202416916 DOI: https://doi.org/10.1002/adfm.202416916
[129] Lai, J., Xiao, L., Zhu, B., Xie, L., Jiang, H.: 3D printable and myoelectrically sensitive hydrogel for smart prosthetic hand control. Microsystems & Nanoengineering 11(1), 15 (2025). https://doi.org/10.1038/s41378-024-00825-y DOI: https://doi.org/10.1038/s41378-024-00825-y
[130] Hui, Y., Yao, Y., Qian, Q., Luo, J., Chen, H., Qiao, Z., Yu, Y., Tao, L., Zhou, N.: Three-dimensional printing of soft hydrogel electronics. Nature Electronics 5(12), 893–903 (2022). https://doi.org/10.1038/s41928-022-00887-8 DOI: https://doi.org/10.1038/s41928-022-00887-8
[131] Yan, Y., Deng, W., Xie, D., Hu, J.: Silk Fibroin Hydrogel for Pulse Waveform Precise and Continuous Perception. Advanced Healthcare Materials 14(4), 2403637 (2024). https://doi.org/10.1002/adhm.202403637 DOI: https://doi.org/10.1002/adhm.202403637
[132] Nezafati, M., Salimiyan, N., Salighehdar, S., Sedghi, R., Dolatshahi-Pirouz, A., Mao, Y.: Facile fabrication of biomimetic and conductive hydrogels with robust mechanical properties and 3D printability for wearable strain sensors in wireless human-machine interfaces. Chemical Engineering Journal 509, 161112 (2025). https://doi.org/10.1016/j.cej.2025.161112 DOI: https://doi.org/10.1016/j.cej.2025.161112
[133] Zhang, Z., Wang, Z., Wang, X., Zhou, X., Zhang, X., Wang, J.: Synthesis of Mimicking Plant Cell Wall-Like Anti-Swelling Hydrogels Based on a ”Bottom-Up” Strategy and Their Application in Osmotic Energy Harvesting. Advanced Functional Materials 35(34), 2502946 (2025). https://doi.org/10.1002/adfm.202502946
[134] Zhang, Z., Wang, Z., Wang, X., Zhou, X., Zhang, X., Wang, J.: Synthesis of Mimicking Plant Cell Wall-Like Anti-Swelling Hydrogels Based on a “Bottom-Up” Strategy and Their Application in Osmotic Energy Harvesting. Advanced Functional Materials 35(34), 2502946 (2025). https://doi.org/10.1002/adfm.202502946 DOI: https://doi.org/10.1002/adfm.202502946
[135] Madika, B., Saha, A., Kang, C., Buyantogtokh, B., Agar, J., Wolverton, C.M., Voorhees, P., Littlewood, P., Kalinin, S., Hong, S.: Artificial Intelligence for Materials Discovery, Development, and Optimization. ACS Nano 19(30), 27116–27158 (2025). https://doi.org/10.1021/acsnano.5c04200 DOI: https://doi.org/10.1021/acsnano.5c04200
[136] Wang, Y., Li, Y., Tang, Z., Li, H., Yuan, Z., Tao, H., Zou, N., Bao, T., Liang, X., Chen, Z., et al.: Universal materials model of deep-learning density functional theory Hamiltonian. Science Bulletin 69(16), 2514–2521 (2024). https://doi.org/10.1016/j.scib.2024.06.011 DOI: https://doi.org/10.1016/j.scib.2024.06.011
[137] Hu, J., Li, Q., Fu, N.: Generative AI for Materials Discovery: Design Without Understanding. Engineering 39, 13–17 (2024). https://doi.org/10.1016/j.eng.2024.07.008 DOI: https://doi.org/10.1016/j.eng.2024.07.008
[138] Zeni, C., Pinsler, R., Z¨ugner, D., Fowler, A., Horton, M., Fu, X., Shysheya, A., Crabb´e, J., Sun, L., Smith, J., et al.: MatterGen: a generative model for inorganic materials design. arXiv preprint arXiv 2312.03687 (2023). https://doi.org/10.48550/arXiv.2312.03687
[139] Merchant, A., Batzner, S., Schoenholz, S.S., Aykol, M., Cheon, G., Cubuk, E.D.: Scaling deep learning for materials discovery. Nature 624(7990), 80–85 (2023). https://doi.org/10.1038/s41586-023-06735-9 DOI: https://doi.org/10.1038/s41586-023-06735-9
[140] Xu, S., Xia, X., Yu, Q., Parakh, A., Khan, S., Megidish, E., You, B., Hemmerling, B., Jayich, A., Beck, K., et al.: 3D-printed micro ion trap technology for quantum information applications. Nature 645(8080), 362–368 (2025). https://doi.org/10.1038/s41586-025-09474-1 DOI: https://doi.org/10.1038/s41586-025-09474-1
[141] Shi, i., Kim, S., Li, P., Dong, F., Yang, C., Nam, B., Han, C., Eig, E., Shi, L.L., Niu, S., et al.: Active biointegrated living electronics for managing inflammation. Science 384(6699), 1023–1030 (2024). https://doi.org/10.1126/science.adl1102 DOI: https://doi.org/10.1126/science.adl1102
[142] Nakamura, K., Di Caprio, N., Burdick, J.A.: Engineered Shape-Morphing Transitions in Hydrogels Through Suspension Bath Printing of Temperature-Responsive Granular Hydrogel Inks. Advanced Materials 36(47), 2410661 (2024). https://doi.org/10.1002/adma.202410661 DOI: https://doi.org/10.1002/adma.202410661
[143] Cao, R., Chen, G., Wang, L., Wang, W., Ni, C., Xia, Z., Hu, A., Hu, W., Lin, H., Qian, P., et al.: 4D Printed Hydrogel Expanders for Personalized and Accelerated Soft Tissue Regeneration. Advanced Materials, e12662 (2025). https://doi.org/10.1002/adma.202512662 DOI: https://doi.org/10.1002/adma.202512662
[144] Ni, C., Zhang, C., Qin, Z., Ke, Q., Wu, B., Zhuoruo, X., Sun, Y., Fu, Q., Chen, D., Zheng, N., et al.: 4D printing of trigger-free shape-memory hydrogels towards self-adaptive substrates for bioelectronics. Nature Communications 17, 677 (2026). https://doi.org/10.1038/s41467-025-67323-1 DOI: https://doi.org/10.1038/s41467-025-67323-1
[145] Lin, W., Zhang, J., Pan, G., Lin, Y., Chen, D., Lu, J., Li, J., Huang, J., Li, Z., Lin, X., et al.: Multifunctional Wide-Temperature Threshold Conductive Eutectic Hydrogel Sensor for Shoulder Pain Monitoring and Photothermal Therapy. Advanced Functional Materials 35(49), e09779 (2025). https://doi.org/10.1002/adfm.202509779 DOI: https://doi.org/10.1002/adfm.202509779
[146] Zhianmanesh, M., Khodaei, A., Crago, M., Lotz, O., Naficy, S., Dehghani, F., Bilek, M., Amin Yavari, S., Akhavan, B.: Universal Method for Covalent Attachment of Hydrogels to Diverse Polymeric Surfaces for Biomedical Applications. Advanced Materials 38(1), e03524 (2025). https://doi.org/10.1002/adma.202503524 DOI: https://doi.org/10.1002/adma.202503524
[147] Ye, X., Chen, Y., Lv, C., Ying, Y., Ping, J., Pan, J., Lan, L.: In situ formed hydrogels for soft bioelectronics. Materials Horizons 12(22), 9537–9555 (2025). https://doi.org/10.1039/D5MH01356A DOI: https://doi.org/10.1039/D5MH01356A
[148] Xu, W., Gu, Y., Xia, W., Yu, A., Fan, W., Qian, S., Shu, Q., Liu, B., Li, Y.: Zwitterionic Surfactant Enhanced Stable Hydrogels for Epidermal Sensors and External Contact Object Perception. ACS Sensors 10(7), 5199–5208 (2025). https://doi.org/10.1021/acssensors.5c01319 DOI: https://doi.org/10.1021/acssensors.5c01319
[149] Wang, J., Xu, Y., Li, S., Tang, L., Li, X., Song, J., Liu, Y.: Physical entanglement improves the anti-adsorption and super-lubricity properties of polyacrylamide-based hydrogels for biomedical applications. Advanced Composites and Hybrid Materials 8, 208 (2025). https://doi.org/10.1007/s42114-025-01267-4 DOI: https://doi.org/10.1007/s42114-025-01267-4
[150] Sun, Q., Hong, K., Fan, L., Zhang, X., Wu, T., Du, J., Zhu, Y.: Bioabsorbable Zwitterionic Hydrogels Achieving Excellent Protein Repulsion and Cell Adhesion. Chemistry of Materials 35(21), 9208–9224 (2023). https://doi.org/10.1021/acs.chemmater.3c01923 DOI: https://doi.org/10.1021/acs.chemmater.3c01923
[151] Gu, Y., Luo, Y., Guo, Q., Yu, W., Li, P., Wang, X., Ye, T., Chang, H., Yuan, W., Wu, H., et al.: Empowering Human-Machine Interfaces: Self-Powered Hydrogel Sensors for Flexible and Intelligent Systems. Advanced Functional Materials, e09085 (2025). https://doi.org/10.1002/adfm.202509085 DOI: https://doi.org/10.1002/adfm.202509085
[152] Kunwar, P., Xiong, Z., Zhu, Y., Li, H., Filip, A., Soman, P.: Hybrid Laser Printing of 3D, Multiscale, Multimaterial Hydrogel Structures. Advanced Optical Materials 7(21), 1900656 (2019). https://doi.org/10.1002/adom.201900656 DOI: https://doi.org/10.1002/adom.201900656
Downloads
Published
Issue
Section
License
Copyright (c) 2026 Authors

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.



