Magnetic Field-Enabled Piezocatalysis: Mechanisms, Materials, and Emerging Biomedical Applications
DOI:
https://doi.org/10.64509/jim.11.66Keywords:
Magnetostrictive Effect, Piezoelectric Effect, Piezocatalytic TherapyAbstract
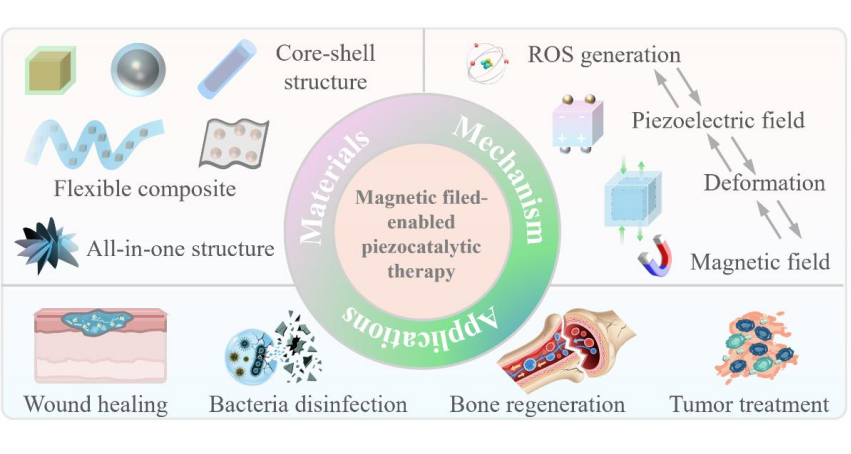
Magnetic field-enabled piezocatalytic therapy represents an emerging frontier in non-invasive biomedical treatments, integrating the principles of magnetostriction, piezoelectricity, and magnetoelectric coupling to remotely drive reactive oxygen species (ROS) generation for therapeutic applications. In this Perspective, we first elucidate the fundamental mechanisms underlying magnetostrictive deformation, piezoelectric polarization, and strain-mediated magnetoelectric coupling, highlighting how mechanical energy derived from magnetic fields can be efficiently converted into localized electric potentials and catalytic activity. We then examine the design and function of magnetostrictive-piezoelectric composite materials, emphasizing core-shell architectures, interface engineering, and nanostructuring strategies that enhance ROS production. Recent advancements in magnetic field-enabled piezocatalytic tumor inhibition, along with comparisons to conventional ultrasound-activated approaches, are discussed. Finally, we outline critical challenges and future directions from the perspectives of material optimization, biocompatibility, mechanistic elucidation, and in vivo evaluation, aiming to guide the rational development of next-generation remotely controllable piezocatalytic therapeutics. This Perspective paper presents a conceptual framework for the design and implementation of magnetic field-enabled piezocatalysis, establishing a connection between materials science and biomedical innovation.
Downloads
References
[1] Nakachi, K., Ikeda, M., Konishi, M., Nomura, S., Katayama, H., Kataoka, T., Todaka, A., Yanagimoto, H., Morinaga, S., Kobayashi, S., et al.: Adjuvant S1 compared with observation in resected biliary tract cancer (JCOG1202, ASCOT): a multicentre, openlabel, randomised, controlled, phase 3 trial. Lancet 401(10372), 195–203 (2023). https://doi.org/10.1016/S0140-6736(22)02038-4
[2] Tarantino, P., Carmagnani Pestana, R., Corti, C., Modi, S., Bardia, A., Tolaney, S.M., Cortes, J., Soria, J.C., Curigliano, G.: Antibody-drug conjugates: smart chemotherapy delivery across tumor histologies. A Cancer Journal for Clinicians 72(2), 165–182 (2022). https://doi.org/10.3322/caac.21705
[3] Zhang, Z., Liu, X., Chen, D., Yu, J.: Radiotherapy combined with immunotherapy: the dawn of cancer treatment. Signal Transduction and Targeted Therapy 7(1), 258 (2022). https://doi.org/10.1038/s41392-022-01102-y
[4] Petersenn, S., Fleseriu, M., Casanueva, F. F., Giustina, A., Biermasz, N., Biller, B. M. K., Bronstein, M., Chanson, P., Fukuoka, H., Gadelha, M., et al.: Diagnosis and management of prolactin-secreting pituitary adenomas: a pituitary society international consensus statement. Nature Reviews Endocrinology 19(12), 722–740 (2023). https://doi.org/10.1038/s41574-023-00886-5
[5] Vellano, C.P., White, M.G., Andrews, M.C., Chelvanambi, M., Witt, R.G., Daniele, J.R., Titus, M., McQuade, J L., Conforti, F., Burton, E.M., et al.: Androgen receptor blockade promotes response to BRAF/MEKtargeted therapy. Nature 606(7915), 797–803 (2022). https://doi.org/10.1038/s41586-022-04833-8
[6] Niknafs, N., Balan, A., Cherry, C., Hummelink, K., Monkhorst, K., Shao, X.M., Belcaid, Z., Marrone, K.A., Murray, J., Smith, K.N., et al.: Persistent mutation burden drives sustained anti-tumor immune responses. Nature Medicine 29(2), 440–449 (2023). https://doi.org/10.1038/s41591-022-02163-w
[7] Wu, Q., Zhang, H., Liu, H.: External physical fielddriven nanocatalytic cancer therapy. BMEmat 1(1), e12010 (2023). https://doi.org/10.1002/bmm2.12010
[8] Ge, M., Xu, D., Chen, Z., Wei, C., Zhang, Y., Yang, C., Chen, Y., Lin, H., Shi, J.: Magnetostrictivepiezoelectric-triggered nanocatalytic tumor therapy. Nano Letters 21(16), 6764–6772 (2021). https://doi.org/10.1021/acs.nanolett.1c01313
[9] Yao, X., Li, D., Gao, C., Deng, Y., Zhang, J., Shuai, C.: Magnetostrictive strain-sensitivity synergy for laserbeam powder bed fusion processed Fe81Ga19 alloys by magnetic field annealing. Advanced Powder Materials 3(5), 100216 (2024). https://doi.org/10.1016/j.apmate.2024.100216
[10] Pan, S., Li, J., Wen, Z., Lu, R.; Zhang, Q., Chen, Y., Wang, S.: Halide Perovskite Materials for Photo (Electro) Chemical Applications: Dimensionality, Heterojunction, and Performance. Advanced Energy Materials 12, 2004002 (2022). https://doi.org/10.1002/aenm.202004002
[11] Gao, C., Zeng, Z., Peng, S., Shuai, C.: Magnetostrictive alloys: Promising materials for biomedical applications. Bioactive Materials 6(10), 3596–3607 (2022). https://doi.org/10.1016/j.bioactmat.2021.06.025
[12] Pascual-Gonz´alez, C., Amor´ın, H., P´erez del Real, R., Serrano-S´anchez, F.J., Palomares, F.J., Castro, A., Alguer´o, M.: Mechanochemically assisted synthesis, inversion degree and magnetization of spinel compounds across the (Zn,Mg)Fe2O4 system for flexible smart devices. Ceramics International 50(24), 53562-53570 (2024). https://doi.org/10.1016/j.ceramint.2024.10.205
[13] Zhou, X., Shen, B., Lyubartsev, A., Zhai, J., Hedin, N.: Semiconducting piezoelectric heterostructures for piezo- and piezophotocatalysis. Nano Energy 96(1), 107141 (2022). https://doi.org/10.1016/j.nanoen.2022.107141
[14] Kwak, W., Yin, J., Wang, S., Chen, J.: Advances in triboelectric nanogenerators for self-powered wearable respiratory monitoring. FlexMat 1, 5–22 (2024). https://doi.org/10.1002/flm2.10
[15] Ye, Y.-Z., Liu, H.-Y., Gong, Y.-J., Xu, Z.-Z., Zhao, Y., Yu, N., Wang, Q.-S., Wen, W., Yang, T.-Y., Li, W., et al.: Design of piezoelectric organic cage salts for energy harvesting. Chem 9(12), 3600–3612 (2024). https://doi.org/10.1016/j.chempr.2023.12.006
[16] Vijayakanth, T., Liptrot, D.J., Gazit, E., Boomishankar, R., Bowen, C.R.: Recent advances in organic and organic-inorganic hybrid materials for piezoelectric mechanical energy harvesting. Advanced Functional Materials 32, 2109492 (2022). https://doi.org/10.1002/adfm.202109492
[17] Ganesha, C., Vijaylakshmi, D.: Magnetoelectric coupling in ferromagnetic/ferroelectric heterostructures: A survey and perspective. Journal of Alloys and Compounds 928, 167181 (2022). https://doi.org/10.1016/j.jallcom.2022.167181
[18] Wei, H., Zhou, L., Cao, F., Chen, Y., Li, B., Kou, J., Lu, C., Xu, Z. : Constructing magnetically propelled piezoelectric and pyroelectric bifunctional micromotors to boost the photocatalytic H2 production involving biomass reforming. Nano Energy 129, 110064 (2024). https://doi.org/10.1016/j.nanoen.2024.110064
[19] Wei, H., Liu, B., Cao, F., Li, B., Kou, J., Chen, Y., Lu, C., Xu, Z.: Spontaneous chirality transition-mediated piezo-pyroelectric synergy for photocatalytic hydrogen production from saturated vapor of VOC aqueous solutions. Advanced Functional Materials e18394 (2025). https://doi.org/10.1002/adfm.202518394
[20] Zhang, Y., Du, D., Fang, C., Yu, X., Fang, Y., Liu, X., Ou, D., Yin, H., Liu, H., Wang, T., et al.: Epigenetics disruptions enabled by porphyrin-derived metal-organic frameworks disarm resistances to sonocatalytic ROS anti-tumor actions. Fundamental Research 5, 296–306 (2025). https://doi.org/10.1016/j.fmre.2022.06.020
[21] Li, Y., Lv, C., Li, Z., Chen, C., Cheng, Y.: Magnetic modulation of lysosomes for cancer therapy. Wiley Interdiscip Rev Nanomed Nanobiotechnol 16, e1947 (2024). https://doi.org/10.1002/wnan.1947
[22] Mendes, M.C., Martins, E.A.G., Chernozem, R.V., Chernozem, P.V., Custodio, C.C., Surmenev, R.A., Kholkin, A.L., Silva, A.S., Mano, J.F.: Osteogenic differentiation triggered by intracellular magnetoelectric stimulation of core–shell nanotransducers under remotely applied magnetic fields. ACS Nano 19(50), 42022–42045 (2025). https://doi.org/10.1021/acsnano.5c10509
[23] Yang, Y., Wen, P., Chen, X., Wang, Y., Zhu, S., Ni, Z., Yuan, L., Shan, L., Zhang, P., Shi, P., et al.: Mechanically-compliant magnetoelectric sutures for wound management. Advanced Functional Materials 35, e71680 (2025). https://doi.org/10.1002/adfm.71680
[24] Wu, L., Liu, Y., Gao, H., Guan, W., Sun, S., Liu, H., Chu, X., Li, G.: In situ non-invasive electrical stimulation by magnetically-actuated piezoelectric Fe₃O₄/PVDF alignment scaffolds for synergistically promoting long-distance peripheral nerve regeneration. Chemical Engineering Journal 509, 161308 (2025). https://doi.org/10.1016/j.cej.2025.161308
Downloads
Published
Issue
Section
License
Copyright (c) 2026 Authors

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.



